Small animal - July 2019
Heart murmurs in young dogs and cats: differentials, tips and additional testing
Ilaria Spalla DVM PhD MRCVS MVetMed DACVIM, veterinary cardiology specialist, Ospedale Veterinario San Francesco (Milan, Italy) provides a comprehensive overview of heart murmurs in young dogs and cats
Heart murmurs can occasionally be auscultated in young dogs and cats; their discovery can be in conjunction with other signs of cardiac disease or it may be an incidental finding. The identification of heart murmurs can cause apprehension for owners, and depending on the location, intensity and characteristics, the veterinary professional can perform a list of differentials and suggest the best diagnostic approach.
Cardiac auscultation
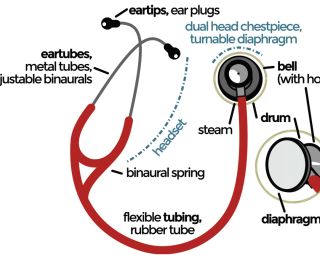
The first stethoscope was invented in 1816 by René Laennec in Paris (Rishniw, 2018) and it has been, since then, a very powerful instrument to aid clinicians in their everyday cardiac evaluation. Auscultation takes time and practice, but it can help in stratifying the risk associated with the identification of an abnormal finding. It is very important that the stethoscope is in good condition too.
In dogs and cats, normal auscultation is characterised by two audible heart sounds (S1 and S2), which separate systole (S1-S2) from diastole (S2-S1). A quiet environment and a relaxed patient provide the best clinical condition to detect abnormalities; however, this sometimes may not happen in everyday practice. Strategies to reduce environmental noise levels and calm the patient, as well as repeated auscultation can be attempted to increase efficacy or confirm the initial suspicion. The patient should be standing, and palpation of the chest walls should be performed prior to applying the stethoscope to identify the precordium and look for thrills, if present (Smith et al, 2006).
Inching (moving the stethoscope between the cardiac apex and base) helps in identifying any abnormal heart sound and its point of maximal intensity (PMI). Traditionally, three areas are heard on the left hemithorax (pulmonary, aortic and mitral [PAM] from a more cranial to caudal position) and one on the right (tricuspid valve [T] [Smith et al, 2006]). Some murmurs may be well localised, whereas some others irradiate to the other hemithorax. Auscultation of the thoracic inlet may aid in identifying particular murmur irradiation (ie. aortic stenosis).
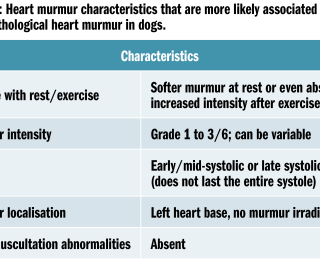
Heart murmurs are the result of turbulent blood flow. The Reynolds number gives the probability of a flow to be laminar (low) or turbulent (high). The Reynolds number has no practical use but is helpful in explaining which conditions can increase the chances of a flow to become turbulent, and therefore to cause a heart murmur (Sisson DD and Ettinger SJ, 1999). These include increased blood velocity, decreased blood viscosity (such as in anaemic patients), changes in vessel/valve radius (such as stenosis, which is generally associated with increased velocity) or changes in blood density. Heart murmurs can either be associated with heart disease (pathological murmur) or may occur in an otherwise normal heart (non-pathological, normal, innocent, functional or physiological murmur). A slight difference in meaning can be given when using the words ‘functional’ or ‘innocent’ murmur. Functional murmurs can be defined as heart murmurs in the absence of structural heart disease, but a plausible explanation for the murmur can be identified (ie. anaemia), whereas an innocent murmur defines a heart murmur in the absence of structural heart disease without any obvious causative explanation (Coté et al, 2015). Non-pathological murmurs may also come with peculiar auscultatory characteristics that can aid in their recognition (see Table 1).
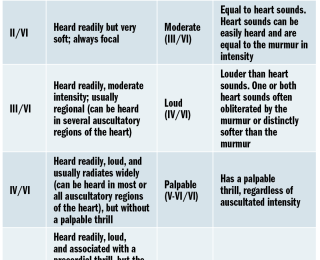
Non-pathological heart murmurs have been identified in young puppies (Szatamari et al, 2015), boxer dogs (Höglund et al, 2004), retired racing hounds (Fabrizio et al, 2006) and whippets (Bavegem et al, 2011), but they can also be auscultated in animals with systemic disease (ie. anaemia, fever).The most common murmur grading scheme proposed in veterinary medicine comes from Ettinger and Suter, a six-level scheme adapted from the Detweiler’s first veterinary grading scheme (see Table 3).
More recently, a simplified grading scheme in dogs with mitral valve disease seemed to provide enough clinical information when only four grading categories were applied (soft – grade I+II/VI; moderate – grade III; loud – grade IV; and thrilling – grade V+VI/VI [Ljungvall et al, 2014]). In dogs with pulmonic stenosis and subaortic stenosis, an even simpler classification was considered to be already informative in discriminating mild disease from moderate/severe disease, the latter of which would require further workup (anything louder than soft murmurs; ie. murmur grades greater than III/VI [Caivano et al, 2018]).
Heart murmur in puppies – differentials
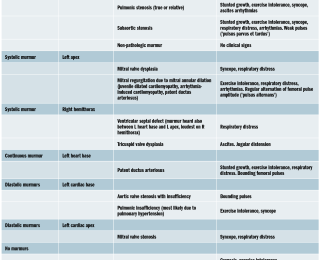
Congenital heart disease (CHD) is probably the number one differential in a young dog with a murmur. Depending on the location and timing, a list of most likely differentials can be drawn (see Table 3).
Most young dogs with murmurs may present without additional clinical signs, but some others may present for exercise intolerance, respiratory effort, ascites or syncopal events. The list of differentials may be different in these two categories of patients, as well as the diagnostic approach. When clinical signs develop, particularly if they lead to the diagnosis of congestive heart failure, it is necessary to treat heart failure prior to performing further investigations.
The prevalence of CHDs in the dog population ranges between 0.13% in mixed-breed dogs from an American shelter (Schrope et al, 2015) to 22% of all cardiac cases in a secondary referral hospital (Oliveira et al, 2011). Congenital heart defects’ prevalence is likely linked to breeds’ prevalence, but the most common simple CHD have not changed much since the first disease prevalence studies were reported in the 1970s. These include pulmonic stenosis, subaortic stenosis and patent ductus arteriosus. Depending on the murmur quality, a list of differentials can be drawn (Table 3).
Some breeds may be more prone to developing specific CHDs, which may not be very common in the overall population. Mitral valve dysplasia is very common in bull terrier dogs (Oliveira et al, 2011), as tricuspid valve dysplasia is for Labrador retrievers (Oliveira et al, 2011).
Some dogs with CHDs may present with clinical signs but may have very soft murmurs or none at all. These CHD are less common but may be worth considering whenever you are assessing a young patient with history of exercise intolerance or syncope. The list of differentials should include Tetralogy of Fallot (most common cyanotic CHD), which can be associated with a left base systolic murmur with different intensity (the softer the more severe the disease), reversed right to left shunting patent ductus arteriosus or ventricular septal defect (both not associated with any heart murmur). Because some of the clinical signs associated with right-to-left shunting are due to hypoxia and polycythaemia, a blood haematocrit is a valid ancillary test to determine the chronicity of the disease process. Atrial septal defects or less common congenital heart defects (cor triatriatum dexter, atrioventricular septal defects to cite a couple) lack heart murmurs.
Another noteworthy differential in a dog with low-grade systolic heart murmur is non-pathological heart murmur. The prevalence of non-pathological murmurs reported in a study of young dogs (one to five years old) varied between 6-12% (Szatamari et al, 2015) and was 16% in another study including only mixed-breed dogs (Schrope et al, 2015). The murmurs were all systolic, low-grade, and mainly at the left cardiac base. Some breeds of athletic dogs may also have a higher prevalence of innocent murmurs (up to 58% in whippets and 67% in retired racing greyhounds [Bavegem et al, 2011] [Fabrizio et al, 2004]). Boxer dogs may present innocent murmurs, but mild subaortic stenosis may also be present or should be excluded by echocardiography (Höglund et al, 2006). Any chamber dilation leading to mitral annular dilation can result in a soft, left apical heart murmur.
Juvenile dilated cardiomyopathy has been identified in Portuguese water dogs (Sleeper et al, 2002). This is associated with an autosomal recessive mutation causing myocardial atrophy. Clinical signs, heart murmurs and auscultation abnormalities tend to develop close to decompensation and death (mean age four months, range two to 32 weeks). A gallop sound (audible S3) may also be auscultated in these patients. Toy Manchester terrier dogs also seem to present a similar form of juvenile dilated cardiomyopathy, with similar sudden decompensation or sudden death (Legge et al, 2013).
Arrhythmia-induced cardiomyopathy (previously known as tachycardia-induced cardiomyopathy) may also be a differential in young dogs with irregular heart rhythms and a soft, left apical murmur. Congenital arrhythmias due to accessory pathways may cause sustained supraventricular tachycardia leading to a dilated cardiomyopathy phenotype and heart murmur due to mitral and/or tricuspid regurgitation (Gopinathannair et al, 2015).
Up to 46.1% of dogs with an accessory pathway had a dilated cardiomyopathy phenotype and 38% of dogs were in congestive heart failure on presentation. Concurrent CHDs were identified in 13% of dogs as well (Wright et al, 2018; Santilli et al, 2018). Labrador retrievers are over-represented, but other breeds are affected as well. Radiofrequency catheter ablation was successful in ablating the accessory pathways in nearly all patients in both centres.
Diet-induced dilated cardiomyopathy has also been recently identified in dogs fed grain-free diets that were not standard breeds affected by dilated cardiomyopathy (Adin et al, 2019); mean age at presentation was four years, but a thorough anamnesis should include the puppy’s diet given these recent findings and the risk of developing diet-induced cardiomyopathy.
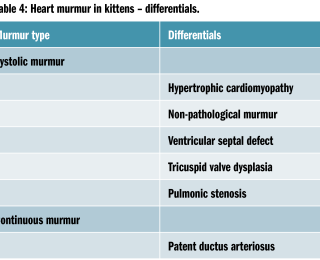
Heart murmur in kittens – differentials
Heart murmurs are common in cats (from 15.5% to 40.8% depending on the study, Payne et al, 2015). It is even more difficult to define the location of a heart murmur in a cat; inching can be performed, and listening at the sternum sometimes can help identifying murmurs in a cat. Purring is one of the most common limitations to cardiac auscultation in a cat; an attempt to stop purring can be made by opening a water tap or gently closing the nose to the cat. Alcohol smell has been anecdotally used, but this is generally unpleasant and can upset some of the cats, so should be avoided. Murmur intensity, timing and variability should be noted. Some murmurs are labile and may not always be present. Whenever a murmur is heard in a cat, an echocardiogram should be suggested to the owner as an additional step to investigate its source, given the high prevalence of non-pathological disease.
The top two differentials in a cat with murmur are hypertrophic cardiomyopathy and non-pathological murmurs, with a nearly 50:50 ratio. Fewer studies investigated the prevalence of CHD in kittens; a prevalence of 0.14-0.5% has been reported (Payne et al, 2015). The most common CHD included ventricular septal defects, tricuspid valve dysplasia, pulmonic stenosis, atrial septal defect or aortic stenosis (Tidholm et al, 2015). Pedigree cats may also have more complex or rare CHD.
Juvenile hypertrophic cardiomyopathy is a very common cause of heart murmur in young cats. Pedigree cats (mainly Ragdolls, Maine Coons, Bengals, Sphynxes and Persians) are reported to be at higher risk, with Ragdoll kittens developing severe disease at a young age – as young as a few months (Silverman et al, 2012; Trehiou-Sechi et al, 2012; Borgeat et al, 2014). Non-pedigree cats can show hypertrophic cardiomyopathy (HCM) as well. Hypertrophic cardiomyopathy can also be present in the absence of a heart murmur and undiagnosed HCM is not an uncommon cause of unexpected death in the feline population (Wilkie LJ et al, 2017).
Systolic anterior motion of the mitral valve leading to left ventricular dynamic outflow tract obstruction is generally responsible for heart murmur in a cat with HCM, but may be present in a minority of cats without obvious or equivocal left ventricular hypertrophy (4.4%; Payne et al, 2015). Dynamic right ventricular outflow tract obstruction is another identified cause of functional murmur (Rishniw et al, 2002).
Further testing
Depending on the presence or absence of clinical signs, the choice of additional diagnostic work-up varies. Echocardiography is probably the best diagnostic test to perform once a heart murmur is heard because it allows identification of the cardiac abnormality, if present, or ruling it out, if not. If the patient is in congestive heart failure, focused thoracic ultrasound to detect B-lines, thoracic or abdominal effusion and to assess chamber size is a valid tool (Boysen et al, 2013). Once the patient is stable or if the patient is asymptomatic, a full echocardiogram can be performed to assess chamber size and function as well as valve morphology or abnormal chamber communications.
Electrocardiography is strongly advised whenever heart rate or rhythm is abnormal on auscultation (tachycardias or bradycardias) to identify the type of the arrhythmia and whether this requires medications. It is also helpful to identify for any sign of concealed accessory pathway (ventricular pre-excitation), although this may be present only in the minority of dogs with accessory pathways (30-48% of all cases). Twenty-four-hour Holter monitoring should be considered should a persistent or intermittent arrhythmia be identified on auscultation.
Thoracic radiographs can provide insights into abnormal lung parenchyma, which may be secondary to heart disease or may be an unrelated cause of tachypnoea/respiratory distress in a dog or cat with a heart murmur. Cats can present a more variable radiographic appearance of cardiogenic pulmonary oedema, as well as a not-so-obvious cardiomegaly, in contrast with dogs, where these findings are more standardised (Guglielmini et al, 2015). Patient triage is important as critically ill patients should not be imaged immediately, but should be stabilised prior to taking radiographs; generally, oxygen administration and mild sedation with butorphanol can be provided in order to allow the patient to settle. Focused thoracic ultrasound can also be a quicker initial way to assess for free fluid that could be drained in order to stabilise the patient enough to perform thoracic radiographs.
Cardiac biomarkers are substances specifically produced by the cardiac cells and released in proportion to cardiac damage (troponin I) or atrial/ventricular stretch (atrial and brain natriuretic peptides; ANP/BNP) (Oyama et al, 2015). Biomarkers such as ANP or BNP are very useful in the context of screening for causes of dyspnoea or in older patients with a heart murmur. The most commonly used is BNP, but due to its short half-life, a fragment of the pro-peptide is analysed (N-terminal pro BNP, NT-proBNP). In a prospective study including mainly Cairn terriers and few other breeds, NT-proBNP concentration was not different between dogs with an innocent murmur compared to those without a murmur (300pmol/L vs 326pmol/L). In contrast, pathological murmurs had a higher median NTproBNP (1102 pmol/L). However, few cases of pulmonic stenosis did not show an increase in NT-proBNP values, resulting in a false positive result (Marinus et al, 2017). Dogs with patent ductus arteriosus showed elevated NT-proBNP, which correlated with radiographic and echocardiographic parameters of cardiac enlargement (vertebral heart score and indexed left ventricular systolic chamber) [Hariu CD et al 2013]).
No studies are available for CHD in relation to cats, but NT-proBNP is higher in cats with cardiomyopathy compared to healthy cats, with more severe heart disease being associated with higher NT-proBNP values. A cut-off of 99 or 100pmol/L allowed good differentiation between healthy cats and those affected by cardiomyopathy. The qualitative NTproBNP snap test currently available has a positive result with an average cut-off of 100pmol/L.
Certain parameters also affect NT-proBNP concentration, including breed and kidney disease. Labrador retrievers and Newfoundland dogs have higher basal values compared to other breeds (Sjöstrand et al, 2014) as well as retired healthy greyhounds (Couto KM et al, 2015). Elevated serum creatinine was also associated with elevated NT-proBNP, but stable kidney disease did not seem to be affecting glomerular excretion of either NT-proBNP and troponin I, indicating no major increase in these values in patients with stable kidney disease (Pelander et al, 2017).
NT-proBNP is also higher in cats with dynamic left ventricular outflow tract and hypertrophic cardiomyopathy who are not in congestive heart failure (Payne JR, unpublished data) compared to cats with similar wall thickness but no dynamic left ventricular outflow tract.
Exercise has also been shown to increase NT-proBNP values (Hunt et al, 2018), so working dogs or any active dog should be tested before any strenuous activity is performed, or if this is not possible, then considering exercise as a possible differential for raised NT-proBNP values and a repeated testing when the dog is at rest should be performed.
Troponins indicate acute myocardial damage; they are therefore, increased in cases of acute myocardial damage (ie. myocardial infarcts, myocarditis, etc.), but are not specific for the underlying cause. The most common assessed troponin is troponin I (TnI), but troponin T has also been measured in cats (Langhorn et al, 2014).
Troponins are increased in acquired heart disease but not in the most common CHDs, where the TnI value is similar to healthy dogs (Spratt DP et al, 2005). Cardiac catheterisation following pulmonary balloon valvuloplasty or transvenous pacemaker implantation was associated with raised post-procedural TnI, in contrast with patent ductus arteriosus closure, which had no significant change from baseline (Shih et al, 2009).
Cats with HCM have variable TnI increase, which is generally mild in asymptomatic cats but slightly higher than in healthy cats (median 0.027 for healthy vs 0.103 for asymptomatic cats, Hori et al, 2018). An elevated TnI value (greater than 0.7ng/ml) was associated with cardiac death in a prospective cohort of cats with hypertrophic cardiomyopathy (Borgeat et al 2014), and similar results were identified in another prospective study (Langhorn et al, 2014), where both TnI and troponin T were of prognostic importance. However, troponins are not disease-specific, and TnI is elevated in other cardiac or systemic disease as well, such as transient myocardial thickening, arrhythmias, bartonellosis and thoracic trauma among others (Novo Matos et al, 2018; Oxford et al, 2018; Joseph et al, 2018; Bartoszuk et al, 2017).
Conclusions
Heart murmurs are the result of turbulent blood flow and can be associated with underlying heart disease, as well as being non-pathological, with different conditions in the dog or cat. Further testing may be necessary to fully rule in/out heart disease, but auscultation is an art that needs practice and time and, in experienced hands, with ideal environmental conditions and compliant patients, can aid in identifying a more pathological vs non-pathological murmur.
- Adin D, DeFrancesco TC, Keene B. Echocardiographic phenotype of canine dilated cardiomyopathy differs based on diet type. J Vet Cardiol 2019; 21: 1-9
- Bartoszuk U, Baron Toaldo M, Pereira N et al. Holter evaluation in cats with symptomatic heart disease and with thoracic trauma. J Vet Intern Med 2017;31:206-207.
- Bavegems VC, Duchateau L, Polis IE et al. Detection of innocent systolic murmurs by auscultation and their relation to hematologic and echocardiographic findings in clinically normal whippets. J Am Vet Med Assoc 2011;238:468-71
- Borgeat K, Sherwood K, Payne JR et al. Plasma cardiac troponin I concentration and cardiac death in cats with hypertrophic cardiomyopathy. J Vet Intern Med. 2014;28(6):1731-7.
- Borgeat K, Casamian-Sorrosal D, Helps et al. Association of the myosin binding protein C3 mutation (MYBPC3 R820W) with cardiac death in a survey of 236 Ragdoll cats. J Vet Cardiol. 2014;16(2):73-80.
- Boysen SR, Lisciandro GR. The use of ultrasound for dogs and cats in the emergency room: AFAST and TFAST. Vet Clin North Am Small Anim Pract. 2013;43(4):773-97.
- Caivano D, Dickson D, Martin M, Rishniw M. Murmur intensity in adult dogs with pulmonic and subaortic stenosis reflects disease severity. J Small Anim Pract. 2018;59(3):161-166
Côté E, Edwards NJ, Ettinger SJ, Fuentes VL et al.Management of incidentally detected heart murmurs in dogs and cats. J Am Vet Med Assoc. 2015;246(10):1076-88. - Couto KM, Iazbik MC, Marín LM et al. Plasma N-terminal pro-B-type natriuretic peptide concentration in healthy retired racing greyhounds. Vet Clin Pathol. 2015; 44(3):405-9
- Fabrizio F, Baumwart R, Iazbik MC et al. Left basilar systolic murmur in retired racing greyhounds. J Vet Intern Med 2006;20:78-82
- Gopinathannair R, Etheridge SP, Marchlinski FE et al. Arrhythmia-Induced Cardiomyopathies: Mechanisms, Recognition, and Management. J Am Coll Cardiol. 2015, 13;66(15):1714-28
- Guglielmini C, Diana A. Thoracic radiography in the cat: Identification of cardiomegaly and congestive heart failure. J Vet Cardiol. 2015;17 S1:S87-101
- Joseph JL, Oxford EM, Santilli RA. Transient myocardial thickening in a Bartonella henselae-positive cat. J Vet Cardiol. 2018;20(3):198-203
- Hariu CD, Saunders AB, Gordon SG et al. Utility of N-terminal pro-brain natriuretic peptide for assessing hemodynamic significance of patent ductus arteriosus in dogs undergoing ductal repair. J Vet Cardiol. 2013;15(3):197-204
- Hori Y, Iguchi M, Heishima Y et al. Diagnostic utility of cardiac troponin I in cats with hypertrophic cardiomyopathy. J Vet Intern Med. 2018;32(3):922-929.
- Höglund K, French A, Dukes McEwan J et al. Low intensity heart murmurs in Boxer dogs: inter-observer variation and effects of stress testing. J Small Anim Pract 2004;45:178e85
- Hunt H, Cave N, Bridges J et al. Plasma NT-proBNP and Cell-Free DNA Concentrations after Prolonged Strenuous Exercise in Working Farm Dogs. J Vet Intern Med. 2018;32(1):135-141
- Langhorn R, Tarnow I, Willesen JL et al. Cardiac troponin I and T as prognostic markers in cats with hypertrophic cardiomyopathy. J Vet Intern Med. 2014;28(5):1485-91.
- Legge CH, López A, Hanna P et al. Histological characterization of dilated cardiomyopathy in the juvenile toy Manchester terrier. Vet Pathol. 2013;50(6):1043-52.
- Ljungvall I, Rishniw M, Porciello F et al. Murmur intensity in small-breed dogs with myxomatous mitral valve disease reflects disease severity. J Small Anim Pract. 2014;55(11):545-50.
- Marinus SM, van Engelen H, Szatmári V. N-Terminal Pro-B-Type Natriuretic Peptide and Phonocardiography in Differentiating Innocent Cardiac Murmurs from Congenital Cardiac Anomalies in Asymptomatic Puppies. J Vet Intern Med. 2017;31(3):661-667.
- Novo Matos J, Pereira N, Glaus T et al. Transient Myocardial Thickening in Cats Associated with Heart Failure. J Vet Intern Med. 2018 ;32(1):48-56
- Oliveira P, Domenech O, Silva J et al. Retrospective review of congenital heart disease in 976 dogs. J Vet Intern Med. 2011;25(3):477-83
- Oxford EM, Giacomazzi FB, Moïse NS, Santilli RA. Clinical and electrocardiographic presentations of transient trifascicular block in three cats. J Vet Cardiol. 2018;20(3):204-212
- Oyama MA.Using Cardiac Biomarkers in Veterinary Practice. Clin Lab Med. 2015;35(3):555-66
Payne JR, Brodbelt DC, Luis Fuentes V. Cardiomyopathy prevalence in 780 apparently healthy cats in rehoming centres (the CatScan study). J Vet Cardiol. 2015;17 S1 1:S244-57 - Pelander L, Häggström J, Ley CJ, Ljungvall I.Cardiac Troponin I and Amino-Terminal Pro B-Type Natriuretic Peptide in Dogs With Stable Chronic Kidney Disease. J Vet Intern Med. 2017; 31(3):805-813
- Rishniw M. Murmur grading in humans and animals: past and present. J Vet Cardiol. 2018 Aug;20(4):223-233
- Rishniw M, Thomas WP. Dynamic right ventricular outflow obstruction: a new cause of systolic murmurs in cats. J Vet Intern Med. 2002;16(5):547-52
- Santilli RA, Mateos Pañero M, Porteiro Vázquez DM et al. Radiofrequency catheter ablation of accessory pathways in the dog: the Italian experience (2008-2016). J Vet Cardiol. 2018;20(5):384-397
- Schrope DP. Prevalence of congenital heart disease in 76,301 mixed-breed dogs and 57,025 mixed-breed cats. J Vet Cardiol. 2015;17(3):192-202
- Silverman SJ, Stern JA, Meurs KM. Hypertrophic cardiomyopathy in the Sphynx cat: a retrospective evaluation of clinical presentation and heritable etiology. J Feline Med Surg. 2012;14(4):246-9
- Fox P, Sisson DD, Moise NS. Textbook of canine and feline cardiology. San Louis: Elsevier; 1999
Sjöstrand K, Wess G, Ljungvall I et al. Breed differences in natriuretic peptides in healthy dogs. J Vet Intern Med. 2014;28(2):451-7 - Sleeper MM, Henthorn PS, Vijayasarathy C et al. Dilated cardiomyopathy in juvenile Portuguese Water Dogs. J Vet Intern Med. 2002;16(1):52-62
- Smith FWK, Keene BW, Tilley LP. Rapid interpretation to heart and lung sounds – a guide to cardiac and respiratory auscultation in dogs and cats. San Louis: Elsevier; 2006
- Spratt DP, Mellanby RJ, Drury N, Archer J. Cardiac troponin I: evaluation I of a biomarker for the diagnosis of heart disease in the dog. J Small Anim Pract. 2005 ;46(3):139-45.
- Shih AC, Maisenbacher HW, Barreirinha A et al. Effect of routine cardiovascular catheterization on cardiac troponin I concentration in dogs. J Vet Cardiol. 2009; Suppl 1:S87-92
- Szatmári V, van Leeuwen MW, Teske E. Innocent Cardiac Murmur in Puppies: Prevalence, Correlation with Hematocrit, and Auscultation Characteristics. J Vet Intern Med. 2015;29(6):1524-8
- Tidholm A, Ljungvall I, Michal J et al. Congenital heart defects in cats: A retrospective study of 162 cats (1996-2013). J Vet Cardiol. 2015 Dec;17 S1:S215-9
- Trehiou-Sechi E, Tissier R, Gouni V et al. Comparative echocardiographic and clinical features of hypertrophic cardiomyopathy in 5 breeds of cats: a retrospective analysis of 344 cases (2001-2011). J Vet Intern Med. 2012 ;26(3):532-41
- Wilkie LJ, Smith K, Luis Fuentes V. Cardiac pathology findings in 252 cats presented for necropsy; a comparison of cats with unexpected death versus other deaths.J Vet Cardiol. 2015 Dec;17 S1:S329
- Wright KN, Connor CE, Irvin HM et al. Atrioventricular accessory pathways in 89 dogs: Clinical features and outcome after radiofrequency catheter ablation. J Vet Intern Med. 2018;32(5):1517-1529.
Which of the following breeds have non-pathological murmurs?
a. Whippet
b. German shepherd
c. Jack Russell terriers
d. Toy Manchester terriers
2. Which of the following congenital heart diseases is NOT associated with a heart murmur?
A. Left-to-right shunting patent ductus arteriosus
B. Pulmonic stenosis
C. Pulmonary hypertension
D. Right-to-left shunting ventricular septal defect
3. Which thoracic area can increase the chance of detecting a heart murmur in a cat with hypertrophic cardiomyopathy?
A. Mitral area
B. Left heart base
C. Sternal
D. Tricuspid area
4. If I have an elevated NT-proBNP result and I want to make sure it is truly elevated, what should I enquire about with the owner?
A. Diet
B. Exercise
C. Probiotic use
D. Treatment with homeopathic drugs
5. Which of the following diseases can increase troponin I concentration?
A. Transient myocardial thickening
B. Hyperthyroidism
C. Patent ductus arteriosus closure via interventional cardiology
D. Asthma