Small animal - June 2019
Cognitive dysfunction in the ageing pet
Clare Rusbridge, professor in veterinary neurology at the School of Veterinary Medicine, University of Surrey, discusses cognitive dysfunction in the ageing pet, reviewing key points in history taking for the older patient, other means of diagnosis, nutraceuticals for the ageing pet and the latest updates on treatment
Like humans, dogs are living longer and, similarly, may not always age successfully. Owners expect good healthcare for their senior pets, quite rightly considered members of the family. Incorporating a cognitive function score in geriatric clinics is a means of identifying dogs predisposed to cognitive dysfunction earlier in the course of this gradually progressive neurological decline allowing for a better understanding of the implication of management strategies.
Introduction
A cognitive function deficit suggests that the animal is unable to perceive ie. unable to make sense of the world around it. It typically occurs in conjunction with other signs associated with damage to the limbic system especially the frontal and prefrontal cortex. There is usually a deterioration of all mental functions including memory, cognition, intellect and personality. Individuals not only lose the ability to process and learn from new information, but also lose existing memories. This makes it harder for them to cope with their environment and leads to increased anxiety and fearfulness. Ageing is the most common predisposing cause of cognitive dysfunction in the aged dog and cat.
What happens in the ageing brain?
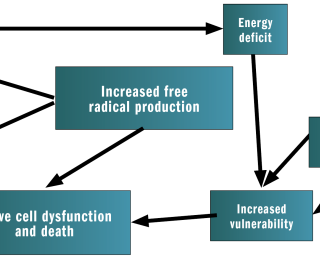
Nerve cells are under constant attack from free radicals produced by normal metabolism (so-called oxidative stress). Free radicals are reactive chemical species with an unpaired electron in atomic or molecular orbit. They damage protein, nucleic acids and membranes containing large quantities of polyunsaturated fat (ie. neuronal cell membranes). The normal defences against oxidative stress are dedicated enzymes and antioxidants such as vitamin E, however these can be easily overwhelmed. The accumulative damage over the lifetime of the animal accounts for the dysfunction and pathology of normal ageing. The ageing brain is under more oxidative stress, is more vulnerable, and is less efficient at coping with it.
Nerve cells are vulnerable to damage by free radicals because of the high lipid content. The ageing cell membrane is even more vulnerable due to a change in structure. The content of the essential fatty acid, docosahexaenoic acid, and vitamin E is significantly decreased and the plasma membrane fluidity is reduced. The aging nerve cell membrane is less efficient at maintaining homeostasis and as a result is more at risk of premature death from excitotoxicity or apoptosis. There are two main sources of free radicals in the aged brain. The first is from inefficient mitochondria. The mitochondria provide energy for all cellular functions. Mitochondria in aged cells fail to produce enough energy and the cell is unable to adequately defend against oxidative attack. The inefficient energy production results in more free radical production further damaging the mitochondria and the cell. The second source is from a toxic protein called beta-amyloid which is deposited in the ageing canine brain (where this is extensive it is referred to as senile or amyloid plaques). Beta-amyloid stimulates an inflammatory response, further free radical production and is neurotoxic.1 A vicious cycle exists because beta-amyloid is aggregated and produces more free radicals in the presence of free radicals. There is a significant association between behavioural dysfunction in dogs and the extent of beta-amyloid deposition.
Hyperphosphorylation of tau is also implicated in cognitive dysfunction following repetitive mild traumatic brain injury (eg. contact sports) frontotemporal dementia and Alzheimer’s disease (tauopathies). Hyperphosphorylated tau with formation of neurofibrillary tangles has been observed in dogs associated with neuroinflammation and cognitive impairment2,3 and also in cats4 and other species. Tau is the major microtubule associated protein (MAP) of a mature neuron. Hyperphosphorylation of tau depresses this biological activity of tau and it is insoluble and unstable. The microtubules disintegrate forming neurofibrillary tangles which interfere with intracellular function.
Identifying dogs that are ageing unsuccessfully
A cognitive function deficit suggests that the animal is unable to perceive ie. unable to make sense of the world around it. It typically occurs in conjunction with other signs associated with damage to the limbic system especially the frontal and prefrontal cortex. The limbic system is involved with behavioural or emotional responses to afferent stimuli eg. smell. There is usually a deterioration of all mental functions including memory, cognition, intellect and personality. Individuals not only lose the ability to process and learn from new information, but also lose existing memories. This makes it harder for them to cope with their environment and leads to increased anxiety and fearfulness. Not surprisingly the first clinical signs of aging are behavioural. Typically, these changes develop insidiously and are often accepted as ‘just getting on a bit’.
Occasionally, signs are precipitated by a sudden change in the dog’s routine, eg. a stay in kennels/hospital.
Clinical signs associated with aging have been summarised by the acronym DISHA, which refers to disorientation, a decrease in social interactions, changes in sleep-wake cycles, a loss of prior housetraining, increased anxiety and changes in their level of activity.5 However, diagnosis can be challenging especially in the early stages of disease because the signs are non-specific and can have alternative explanations.
Common clinical signs
1) Stereotypical pacing
The dog relentlessly and aimlessly paces round and round, typically in the evening or night. They can appear distressed and the owner typically complains that they ‘won’t settle’.
2) Aimless, repeated barking
3) Excessive daytime lethargy
The dog is disinterested in playing, walking or other previously enjoyable pursuits.
4) Decreased social interaction
The owner no longer receives an effusive greeting. The dog seems to exist in isolation without being part of a family.
5) Loss of learned behaviour, eg. housetraining
The dog becomes disinhibited about defecation. The owner may comment that the dog ‘goes in the house even immediately after being outside’. It is important to establish that the dog does not have another reason for incontinence. With cognitive function syndrome the dog will still posture to eliminate.
6) Anxiety
The owner may describe unexplained ‘panic attacks’ or fear of familiar situations/people.
7) Disorientation
The dog may seem to be lost in familiar places, for example, unable to find their way in from the garden. The dog may also lose or run off from the owner on a walk.
8) Variable other neurological deficits (depending on underlying pathology)
The most common are weakness, proprioceptive deficits and becoming stuck in corners and furniture because of an inability to walk backwards/avoid the obstacle. Some neurodegenerative disease characterised by clinical signs of cognitive function syndrome develop a central blindness or deafness for example Canine Lafora’s disease.
The behavioural changes are difficult for the owners to come to terms with. The dog is ‘a shell’ of its previous self and there is mourning for this loss. The relentless pacing at night leads to the conclusion that the dog is unhappy. Defecation and urination in the house is not only distressing but can cause family arguments. Many dogs are euthanised but for most owners, this is not a simple decision. It is far easier to sign away your pet’s life if it is dying of heart disease or cancer.
Differential diagnosis
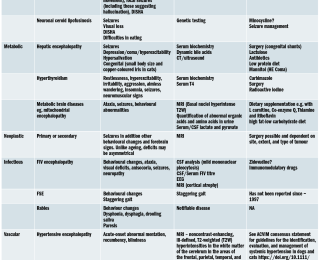
Other conditions affecting the prefrontal cortex will produce similar signs; the most important is a meningioma originating from the olfactory groove or frontal lobe. This is a slow-growing tumour and, typically, older dogs are affected. As a general rule, it is exceptional to see seizures as well as a cognitive function syndrome. An asymmetrical postural deficit, eg. if the dog was hemiparetic or had unilateral proprioceptive deficits, then a mass should be suspected. If the dog were circling in one direction rather than aimlessly wandering, a mass (same side as direction of circling, opposite side to postural deficits) should be considered. Sometimes it can be impossible to differentiate on clinical grounds and MRI is required. Behavioural problems that are not due to organic brain disease should be ruled out.
Work up
History
As the key and early features of the disease are behavioural, taking the time to obtain a history and asking the right questions are paramount. As the decline with ageing develops insidiously, it would be appropriate to ask questions about interaction with the owner, orientation, sleep pattern and learned behaviour as part of a general heath check-up, eg. at booster vaccination or in a nurse’s ‘senior clinic’.
While taking a history, allow the dog off the lead in the consulting room. A dog with rostral forebrain pathology often paces round the consulting room oblivious of furniture and does not acknowledge the vet’s presence. However, remember that this is not a definite sign of ageing and will occur with other serious brain disease. Use of validated questionnaires can help eg. the canine dementia (CADES) cognitive function score.6
Neurological examination
A full neurological examination should be performed. From a differential diagnosis point of view the main aim is the following.
a) If there are deficits, can these be related to disease of the forebrain?
- eg. Behavioural changes (as overleaf)
Seizures - Depression/stupor
- Circling (towards side of lesion)
- Postural deficits (contralateral to lesion)
- Visual deficits (contralateral to lesion, normal pupillary light responses)
b) Is there multifocal disease?
Are there deficits relating to pathology of more than one area of the nervous system? This would either suggest an inflammatory process or a multifocal tumour such as lymphoma. For example vestibular disease or cranial nerve deficits suggest brain stem disease. Hyperaesthesia, hypermetria or an intention tremor suggests cerebellar disease. Central nervous system (CNS) inflammatory disease (and occasionally lymphoma) is typically associated with spinal pain.
Laboratory tests
Systolic blood pressure
Other organ dysfunction and other metabolic disease eg. hypothyroidism should be ruled out. Assessment and management of other comorbidities is important and a recent study looking at the physical examination and laboratory tests in senior and geriatric dogs that were judged by the owner to be healthy found that 32/100 dogs had increased serum creatinine, 29/100 hypophosphatemia, 27/100 increased ALP, 25/100 increased ALT, and 23/100 leukopenia, while 62/96 dogs had crystalluria (mostly amorphous crystals). Overt proteinuria and borderline proteinuria were detected in 13 and 18 of 97 dogs, respectively. Body temperature, haematocrit, serum albumin, and serum total thyroxine concentration were significantly lower in geriatric compared with senior dogs and although stiffness or lameness was reported by few owners, abnormalities on orthopaedic examination were commonly encountered.7
MRI
Cortical atrophy is the hallmark of canine cognitive function syndrome. Due to the loss of brain tissue the CSF filled spaces of the brain, ie. the ventricles and the subarachnoid space, look wider. There is a reduction in the height of the inter-thalamic adhesion (guide 3.82 +/-0.79mm in dogs with CCD; 6.79 +/-0.70mm with normal). MRI changes associated with amyloid deposition in cerebral blood vessels (Cerebral amyloid angiopathy; CAA) are characterised by leukoaraiosis (bilaterally symmetrical periventricular areas of T2 and FLAIR hyperintensity). The damaged blood vessels are predisposed to bleeding. Microbleeds are best demonstrated by MRI with susceptibility weighting, eg. gradient echo. CAA also predisposes ischaemic and haemorrhagic stroke.
Cortical atrophy is not confined to cognitive function syndrome and may be seen in other neurodegenerative diseases however this finding in an aged dog with appropriate clinical signs is sufficient for diagnosis and treatment. Brain biopsy would be required for confirmation. There are many instances where MRI is not possible. In these cases it would be appropriate to start symptomatic therapy and re-evaluate the neurological examination after a few weeks. Progressive neurological deficits, especially if asymmetric, would suggest other progressive neurological disease.
Managing brain ageing
The neurological decline (cognitive impairment) associated with ageing can be treated in two ways.
1) Altering neurotransmission
Signs of brain ageing are, in part, due to neurotransmitter imbalance (ie. the chemical messages, which ‘switch nerve cells on or off’).
a) L-deprenyl (selegiline) which is marketed as Selgian is a monoamine oxidase inhibitor, the enzyme that breaks down the neurotransmitters noradrenalin and dopamine. Cognitive function disorders such as Alzheimer’s have increased monoamine oxidase activity and a monoaminergic transmission defect which selegiline reverses. Selegiline also has neuroprotective functions. It increases antioxidant enzyme activities in selected brain regions and increases nitric oxide in brain tissue and cerebral blood vessels. Nitric oxide modulates many activities including cerebral blood flow and memory and is depressed in brains from patients with cognitive function disorders such as Alzheimer’s studies indicate that Selegiline improves memory and longevity in aged dogs.
Selegiline is dosed once daily at 0.5mg/kg in dogs and 1mg/kg in cats. Although an improvement can be noted within three weeks, it can take up to six weeks for a benefit to be realised (if there is one). All animals should be monitored with serum biochemistry and haematology before and during treatment (every six months). Transient gastrointestinal signs may be reported.
b) Cholinesterase inhibitors eg. tactrine, donepezil and rivastigmine are drugs licenced for treating Alzheimer’s disease. In Alzheimer’s there is a reduction of cholinergic cells in the basal forebrain and these agents prolong the availability of acetylcholine at neuronal synapses. Although they improve cognitive function they do not prevent disease progression Donepezil (1.5mg/kg) has been used experimentally in aged laboratory beagles with a positive benefit.8
2) Neuroprotection eg. altering the nerve cell environment so that they are less likely to be damaged and/or more likely to able to repair.
a) Propentofylline increases the molecular signal adenosine by inhibiting uptake and the breakdown enzyme phosphodiesterase. Although originally thought to be useful by improving cerebral blood flow, it is probably most beneficial by influencing glial cell function. Adenosine stimulates astrocytes - cells responsible for protecting nerve cells from the effects of inflammatory mediators, free radicals and the excitatory neurotransmitter glutamate. Conversely, functions of the potentially neurotoxic microglial cells (the inflammatory cells of the CNS) are depressed.
b) Dietary manipulation/nutraceuticals.
- Lifelong maintenance of lean body mass9 is neuroprotective.
- Dietary antioxidant supplementation. Antioxidant enzymes are significantly reduced in the aged canine brain. Subclinical deficiency of vitamin E and C is a known risk for developing cognitive impairment and supplementing these vitamins and other naturally occurring antioxidants eg. carotenoids and flavonoids can protect against age-related neuronal impairments.
- L-Carnitine plays a regulatory role in mitochondrial transport of long chain fatty acids. It protects against oxidative stress by preserving energy production and has free radical scavenging properties. It has been shown to enhance learning capacity and reduce progressive neurological decline in ageing rats.
- Alpha lipoic acid is a potent free radical scavenger and has been shown to reverse age-related changes and improve memory in aged rodents.
- Supplementing aged animals with essential fatty acids such as gamma linoleic acid and docosahexaenoic acid has been shown to restore neuronal membrane structure and function.
- Aged dogs fed a diet (Hills BD™) supplemented with vitamins E and C, L-Carnitine, lipoic acid, flavonoids, carotenoids, gamma linoleic acid and docosahexaenoic acid were shown to have improved learning ability and memory. There are many nutraceutical products available that contain these or similar antioxidants.
- Medium-chain TAG diet (5.5% MCT eg. Purina Neurocare™) provides the brain with an alternative energy source (ketones) and in aged laboratory beagles improved cognitive function.10
- Ginkgo biloba leaf extract contains many flavone glycosides and terpenoides. It is a free radical scavenger, has a relaxing effect on vascular walls, antagonises platelet activating factors, improves microcirculation and affects neurotransmitters. It has been shown to protect neuronal cell membranes from free radical damage in vitro, improves short-term memory in aged mice and humans and has a positive effect on dementia in humans.
3) Other strategies
a) Behavioural enrichment – lifestyle factors, such as engaging in physical, intellectual, and social activities, are protective against dementia in aged humans and there is evidence of the same in dogs.11 Common strategies are outdoor walks (even if in a stroller); food mazes and puzzles; hiding food; reward-based obedience and simple tricks (using simple unambiguous cues and clear reward signals); encouraging play. Lifelong learning may also be protective.12 In many cases, house training will need to be restarted as if for a puppy. Retraining should be re-established promptly to discourage inappropriate learning. Games, training and exercise should be of short duration as older dogs have reduced concentration and repeated frequently to increase interest (e.g. short walks often).
b) Melatonin (plant form phytomelatonin) plays a major role in maintaining circadian rhythm and it has been suggested that changes in melatonin metabolism might contribute to circadian rhythm disturbances seen in patients with cognitive function disorder. It is an indolamine derived from tryptophan with chronobiotic, antioxidant, anti-inflammatory, neuroprotective, and analgesic properties. Melatonin secretion progressively declines during the normal aging process. A significant reduction of circulating melatonin has also been observed with neurodegenerative disorders such as Alzheimer's disease.13,14 Giving 3mg of Melatonin at bedtime (repeated 2x if necessary) can be helpful in some dogs with sleep disorders.
c) Non-steroidal anti-inflammatory drugs. It has been postulated that these drugs may be useful because amyloid deposition induces an inflammatory response. There is evidence that humans using NSAIDs are at reduced risk of developing cognitive function disorders such as Alzheimer’s disease.
4) Experimental/undergoing trial in laboratory dogs
d) Beta-amyloid (Aβ) immunotherapy (experimental studies only) – an active vaccine against fibrillar Aβ 1–42 (VAC) in aged laboratory beagles resulted in maintenance but not improvement of cognition along with reduced brain Aβ. However, needed to be combined with behavioural enrichment.15
e) Rapamycin, a macrolide immunosuppressive agent and mTOR inhibitor.16
f) Metformin, a biguanide antihyperglycemic agent (anti-diabetes treatment).16
General notes on treatment
In most situations, the improvement takes time and owners should be warned that results are not instantaneous. Often, at least four weeks of therapy is required, hence is more likely to be effective if the problem is identified early. If the owner cannot cope with one more accident on the floor then it is too late. The author uses dietary management alone for very mild cases and dogs at risk. Dogs with more serious deficit have dietary supplementation with one of the above drugs (usually selegiline). If the patient shows an initial response and then deteriorates then a second drug is added (eg. a combination of dietary supplementation, selegiline and vivitonin). Each treatment regime should ideally be given four weeks to assess its effectiveness before making changes.
Conclusion
Neurological dysfunction as a consequence of ageing is common and not inevitable. Early recognition of signs and appropriate management can improve the quality of life of our patients and increase the enjoyment that our clients have from their pets.
- Rusbridge C, Salguero FJ, David MA, et al. An aged canid with behavioural deficits exhibits blood and cerebrospinal fluid amyloid beta oligomers. Frontiers in aging neuroscience 2018;10:7.
- Schmidt F, Boltze J, Jager C, et al. Detection and Quantification of beta-Amyloid, Pyroglutamyl Abeta, and Tau in Aged Canines. Journal of neuropathology and experimental neurology 2015;74:912-923.
- Smolek T, Madari A, Farbakova J, et al. Tau hyperphosphorylation in synaptosomes and neuroinflammation are associated with canine cognitive impairment. The Journal of comparative neurology 2016;524:874-895.
- Chambers JK, Tokuda T, Uchida K, et al. The domestic cat as a natural animal model of Alzheimer's disease. Acta Neuropathol Commun 2015;3:78.
- Chapagain D, Range F, Huber L, et al. Cognitive Aging in Dogs. Gerontology 2018;64:165-171.
- Madari A, Farbakova J, Katina S, et al. Assessment of severity and progression of canine cognitive dysfunction syndrome using the CAnine DEmentia Scale (CADES). Applied animal behaviour science 2015;171:138-145.
- Willems A, Paepe D, Marynissen S, et al. Results of Screening of Apparently Healthy Senior and Geriatric Dogs. Journal of veterinary internal medicine/American College of Veterinary Internal Medicine 2017;31:81-92.
- Araujo JA, Greig NH, Ingram DK, et al. Cholinesterase inhibitors improve both memory and complex learning in aged beagle dogs. J Alzheimers Dis 2011;26:143-155.
- Adams VJ, Watson P, Carmichael S, et al. Exceptional longevity and potential determinants of successful ageing in a cohort of 39 Labrador retrievers: results of a prospective longitudinal study. Acta veterinaria Scandinavica 2016;58:29.
- Pan Y, Larson B, Araujo JA, et al. Dietary supplementation with medium-chain TAG has long-lasting cognition-enhancing effects in aged dogs. The British journal of nutrition 2010;103:1746-1754.
- Head E, Nukala VN, Fenoglio KA, et al. Effects of age, dietary, and behavioral enrichment on brain mitochondria in a canine model of human aging. Experimental neurology 2009;220:171-176.
- Chapagain D, Viranyi Z, Wallis LJ, et al. Aging of Attentiveness in Border Collies and Other Pet Dog Breeds: The Protective Benefits of Lifelong Training. Front Aging Neurosci 2017;9:100.
- Karasek M. Melatonin, human aging, and age-related diseases. Exp Gerontol 2004;39:1723-1729.
- Ahn JH, Park JH, Kim IH, et al. Comparison of arylalkylamine N-acetyltransferase and melatonin receptor type 1B immunoreactivity between young adult and aged canine spinal cord. Journal of veterinary science 2014;15:335-342.
- Davis PR, Giannini G, Rudolph K, et al. Abeta vaccination in combination with behavioral enrichment in aged beagles: effects on cognition, Abeta, and microhemorrhages. Neurobiol Aging 2017;49:86-99.
- Kaeberlein M, Creevy KE, Promislow DE. The dog aging project: translational geroscience in companion animals. Mammalian genome : official journal of the International Mammalian Genome Society 2016;27:279-288.
1. The most common signs in an ageing dog with cognitive dysfunction are:
a. Anxiety, increased daytime lethargy with evening restlessness and repetitive vocalisation
b. Circling with tendency for seizures
c. Displaying aggression towards familiar people
d. Inappropriate urination and/or defecation in the sleeping area
2. Signs suggesting forebrain disease are:
a. Tetraparesis with cranial nerve deficits
b. Circling with contralateral postural and visual deficits
c. Head tilt and vestibular ataxia
d. Blindness with dilated pupils unresponsive to light
3. Dietary supplements useful for managing the early stages of cognitive dysfunction are:
a. Glucosamine HCL, Chondroitin Sulphate, Vitamin C, Zinc Sulphate
b. L-Tryptophan, Gamma-Aminobutyric Acid, L-Theanine, Inositol, Vitamin B6, B3, B1, B12
c. Vitamin E and C, L-Carnitine, lipoic acid, flavonoids, carotenoids, gamma linoleic acid and docosahexaenoic acid
d. Boswellic Acids, Silymarin, Proanthocyanidins
4. MRI signs which would not support a diagnosis of age-associated cognitive function decline are:
a. Leukoaraiosis
b. Decreased height of the intra-thalamic adhesion
c. Cortical atrophy
d. Abnormal uptake of gadolinium contrast
5. Positive Response to selegiline (if there is going to be one) is typically seen within
a. 1 week
b. 2 weeks
c. 4 weeks
d. 6 weeks
Answers: A, B, C, D, D