Surgery of the small intestine in dogs: principles and surgical techniques
In this article, Ronan A. Mullins MVB DECVS PGDipUTL DVMS MRCVS, EBVS European Specialist in Small Animal Surgery, and Associate Professor and Discipline Head of Small Animal Surgery at University College Dublin, provides an overview of the principles of intestinal surgery and selected surgical techniques relevant to small intestinal surgery in dogs
Surgical conditions affecting the small intestine are commonly encountered in small animal practice. Indications for surgery include foreign bodies, intussusception, neoplasia, trauma, and intestinal perforation. Regardless of the underlying condition, successful outcomes depend on adherence to fundamental surgical principles that optimise tissue healing, minimise complications, and preserve intestinal function.
Despite advances in diagnostic imaging and perioperative care, complications such as dehiscence and septic peritonitis remain significant concerns following intestinal surgery. Reported rates of enteric leakage after enterotomy or intestinal resection and anastomosis (R&A) range from approximately seven to 16 per cent, and these complications are associated with high morbidity and mortality.
This article reviews the anatomy of the small intestine, the principles of intestinal surgery, and selected surgical techniques related to small intestinal surgery in dogs.
Preoperative considerations
Appropriate patient stabilisation is essential prior to anaesthesia and surgery. Many dogs present with dehydration, electrolyte imbalances, and acid-base disturbances, particularly in cases of vomiting or obstruction. Intravenous fluid therapy should be instituted to restore circulating volume and correct any deficits. Patients with suspected septic peritonitis or systemic compromise may require more aggressive resuscitation, including broad-spectrum antimicrobial therapy and careful haemodynamic monitoring. Analgesia should be provided early, typically using opioids, with additional agents as required. Antimicrobial therapy is indicated in cases where intestinal compromise, perforation, or contamination is suspected.
Anatomy of the small intestine
A thorough understanding of small intestinal anatomy is critical. The small intestine extends from the pylorus to the ileocolic junction and consists of three segments: the duodenum, jejunum, and ileum. Anatomically, the duodenum is relatively fixed, making surgery in this area more challenging, whereas the jejunum is highly mobile. The descending duodenum begins at the pylorus and travels caudally along the right side of the abdomen within the mesoduodenum. The major duodenal papilla receives the common bile duct and pancreatic duct, while the minor duodenal papilla receives an accessory pancreatic duct. Near the pelvic inlet, the duodenum turns medially, forming the caudal duodenal flexure, before continuing cranially as the ascending duodenum. The initial part of the ascending duodenum is attached to the descending colon by the duodenocolic ligament, an important surgical landmark that limits exteriorisation of the duodenum.
The jejunum forms the largest component of the small intestine and is suspended from the dorsal body wall by the mesentery (mesojejunoileum). This is a large, curtain-like structure that contains the majority of the small intestine, as well as blood vessels, nerves, and lymphatics supplying the small intestine. The ileum is the short, terminal segment of the small intestine. Intraoperatively, it can be identified by its proximity to the caecum and the presence of the antimesenteric ileal vessel, which courses from the caecum toward the jejunum. In contrast, the remainder of the small intestine lacks an antimesenteric vessel, receiving the entirety of its blood supply from the mesenteric side. The ileum enters the ascending colon at the ileocolic orifice.
The small intestine receives its blood supply from the cranial mesenteric artery, forming arcades within the mesentery. From these arcades, short vasa recta supply the intestinal wall. Venous drainage is via the gastroduodenal vein (which drains the proximal duodenum) and cranial mesenteric vein (which drains the distal duodenum, jejunum, and ileum), which ultimately drain into the portal vein.
Histologically, the intestinal wall is composed of four layers: the mucosa, submucosa, muscularis, and serosa. The mucosa is arranged into villi, which increase the absorptive surface area of the intestine. The submucosa is the primary strength-holding layer of the gastrointestinal tract and is, therefore, the critical layer incorporated during intestinal closure.
Surgical approach
The most common approach for small intestinal surgery is via ventral midline celiotomy. The necessary equipment for such surgery includes: a self-retaining retractor such as a Balfour or Gosset; a suction unit, tubing, and Poole suction tip; laparotomy sponges and regular 4 x 4 inch surgical swabs/sponges (with radiographic marker); non-traumatic Doyen intestinal forceps; monopolar and bipolar electrosurgical devices. Following opening into the abdominal cavity, the falciform ligament is dissected from the body wall with monopolar electrosurgery and ligated cranially before excising with scissors (Figure 1).
Moistened laparotomy sponges are placed along either side of the incision to reduce contamination and tissue trauma. Self-retaining abdominal retractors, such as a Balfour, are used to maintain exposure of the peritoneal cavity. A systematic exploration of the entire abdominal cavity should be performed before any definitive procedure. However, if there is an actively leaking intestinal segment, it should be packed off with laparotomy sponges and isolated from the remainder of the abdominal cavity until the exploration is complete. Free peritoneal fluid should be suctioned, and a sample submitted for cytology and culture if septic peritonitis is suspected.
Antibiotic prophylaxis
The small intestine normally contains both Gram-positive and Gram-negative bacteria that are contained by the mucosal barrier. When this barrier is disrupted by disease or surgical manipulation, bacterial contamination of the abdominal cavity may occur. The necessity of perioperative antibiotic administration in intestinal surgery remains debated. Surgery involving entry into the small intestinal lumen is classified as clean-contaminated. If intraoperative leakage of intestinal contents occurs, it is classified as contaminated. Finally, if there is pre-existing perforation and septic peritonitis, the surgery is classified as dirty/infected. Perioperative antimicrobials are typically administered to dogs undergoing clean-contaminated small intestinal surgical procedures, however, postoperative antimicrobial use is not advised. A commonly used protocol at the author’s institution involves administration of amoxicillin-clavulanate or cefuroxime (20 mg/kg IV) 30 minutes preoperatively, with repeat dosing every 90 minutes intraoperatively if the procedure is prolonged. Postoperative antimicrobials are not indicated following clean-contaminated procedures, as they have not been shown to reduce infection rates and may contribute to antimicrobial resistance. Their use is appropriate in contaminated and dirty/infected cases.
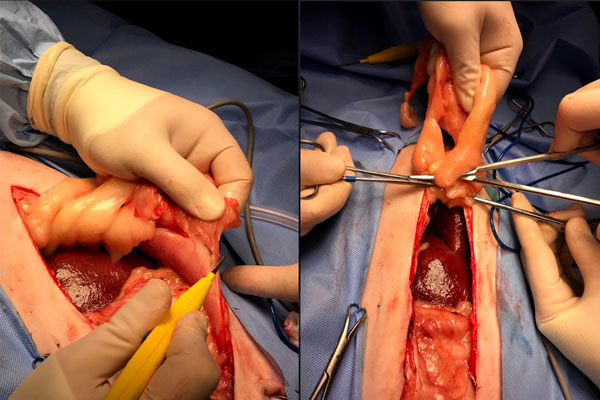
Figure 1: Intraoperative images of a typical ventral midline coeliotomy. The dog’s head is positioned top left (left image) and top (right image). The falciform ligament and fat are visible and dissected from the body wall with monopolar electrosurgery. The tissues are clamped at the level of the xiphoid and an encircling ligature of 3-0/2-0 (depending on the size of the animal) polydioxanone is applied to prevent bleeding.
Determining intestinal viability
Assessment of intestinal viability remains largely subjective. Surgeons typically rely on several criteria, including: serosal surface colour, serosal capillary perfusion, presence of peristalsis, and palpable arterial pulsation in mesenteric vessels. However, these indicators are not always reliable. When intestinal viability is uncertain, resection is often the safest option.
Key Halsted’s Principles: tissue handling, preservation of blood supply, and asepsis
Gentle tissue handling is a critical component of intestinal surgery. The intestinal wall is delicate and easily traumatised, and excessive or repeated manipulation can impair healing. The intestine should not be grasped directly during enterotomy closure or anastomosis; instead, closed DeBakey thumb forceps should be used to gently stabilise the tissue during suture placement. Preservation of the segmental blood supply is essential during resection and anastomosis, as disruption may compromise perfusion and increase the risk of dehiscence. Accurate apposition of the submucosal layer is also crucial for intestinal healing. Strict aseptic technique is essential, particularly in contaminated cases. Measures to minimise intra-abdominal contamination, including isolation of the affected segment and packing with moistened laparotomy sponges, are important to reduce leakage.
Choice of suture material for enteric closure
Monofilament absorbable sutures such as polydioxanone (PDS) are commonly recommended for intestinal closure. Compared with multifilament sutures, monofilament sutures produce less tissue drag, reduce bacterial adherence, and provoke less inflammatory reaction. All suture materials produce an inflammatory reaction after implantation because of their foreign nature and the mechanical disruption of tissue that occurs with their placement. Although multifilament sutures have been used successfully in intestinal surgery, their braided structure may promote bacterial colonisation in contaminated environments.
A swaged-on, taper-point needle should be used for intestinal surgery.
Suture pattern
Successful intestinal healing depends on accurate apposition of the submucosa, which provides the primary strength of the intestinal wall. Good submucosal apposition results in primary intestinal healing with direct bridging of the defect, whereas poor submucosal apposition results in healing by second intention. The intestine is closed in a single layer. Tissues should be well opposed without being crushed. Approximating suture patterns are therefore preferred, including simple continuous, simple interrupted, and modified Gambee. The author most commonly uses a simple continuous suture pattern for enterotomy and anastomosis closure. Both simple interrupted and continuous suture patterns have been shown to result in low and comparable rates of enteric leakage for enterotomy and anastomosis closure in dogs. Suture bites are typically placed full-thickness, approximately 3mm apart and 3mm from the cut edge.
Reinforcement of intestinal closure
Omentalisation
The omentum plays a valuable role in intestinal surgery due to its vascular, lymphatic, and immunologic properties. Often referred to as the ‘policeman of the abdomen’, the omentum can help control infection, improve vascularisation, and promote healing. Although the omentum may naturally migrate to areas of inflammation, intentional placement around an at-risk enterotomy or anastomosis site can easily be performed to try to reduce the risk of dehiscence and leakage. The technique involves wrapping the omentum around the affected intestinal segment and securing it in place with one or two simple interrupted sutures of polydioxanone (PDS) (Figure 2).
When wrapped around an anastomosis, it forms an adherent sheath capable of preventing perforation and fatal leakage while promoting revascularisation.
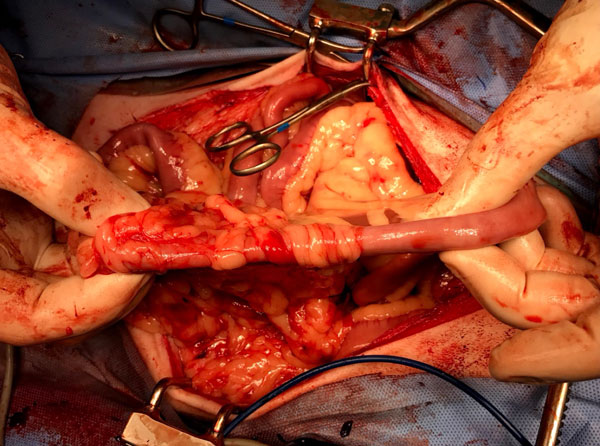
Figure 2: Omentalisation of a jejunal enterotomy site in a dog. The omentum is wrapped around the affected intestinal segment and may be secured in place with one or two simple interrupted polydioxanone (PDS) sutures.
Serosal patching (“the surgical parachute”)
Serosal patching involves suturing a loop of healthy intestine over an at-risk intestinal repair, without tension or creation of any kinks. The technique has been referred to as the ‘surgical parachute’ because of its ability to reinforce intestinal repairs (e.g., anastomoses), and seal contaminated and grossly infected intestinal perforations in dogs. The antimesenteric border of the jejunum is placed longitudinally (in the case of an enterotomy) or transversely (in the case of an anastomosis) over the closure site and secured with interrupted sutures of 3-0/4-0 polydioxanone incorporating the submucosa of both intestinal segments (Figure 3). Although retrospective studies suggest that serosal patching may not significantly improve survival in dogs with septic peritonitis, this likely reflects selection bias, as dogs undergoing serosal patching probably had a greater baseline risk of a poor outcome. In one study, although the difference did not reach statistical significance, dehiscence occurred in 3/18 (16.7 per cent) dogs that received serosal patching compared with 19/64 (29.7 per cent) dogs that did not. This technique may be preferred over omentalisation in dogs with recognised risk factors for dehiscence, including anaemia, hypoalbuminaemia, prior blood transfusion, and preoperative septic peritonitis.
Intestinal leak testing
Leak testing is used to assess the integrity of enteric suture lines prior to abdominal closure; however, one study demonstrated no association with a reduced incidence of postoperative anastomotic dehiscence. The most commonly employed technique involves inflation of an approximately 10cm segment of intestine centred over the suture line with saline, followed by inspection for leakage. To perform this, both ends of the intestinal segment are occluded using digital pressure (assistant’s index and middle fingers) (Figure 4) or non-crushing forceps (e.g., Doyen). A 25-gauge hypodermic needle is inserted into the isolated lumen, and a volume of saline sufficient to achieve moderate distension is injected. While maintaining occlusion, the segment is gently compressed between the surgeon’s thumb and index finger, and the suture line is observed for leakage. It should be recognised that any anastomosis can be made to leak with excessive pressure; therefore, only moderate, physiologic distension should be applied. Air insufflation is a more recently described technique, analogous to testing a bicycle tube for a leak. It involves submerging the occluded intestinal segment in sterile saline and insufflating it with air via a needle attached to a syringe, with observation for escaping bubbles.
An alternative method involves milking intestinal contents across the suture line; however, this is not feasible in an empty bowel and does not replicate physiologic intraluminal pressures, and is therefore not commonly performed.
Selected small intestinal surgical techniques
Enterotomy
Enterotomy is commonly performed for removal of intestinal foreign bodies or collection of biopsy samples. The affected intestinal segment is isolated from the abdominal cavity using laparotomy sponges, and the intestine on either side of the enterotomy occluded with an assistant’s fingers. An incision is made along the antimesenteric border with a no. 11 blade and extended with Metzenbaum scissors. At the ileum, the antimesenteric vessel should be avoided. When removing a foreign body, the incision should be made aboral (distal) to the obstruction in the healthiest section of intestine (Figure 5). In cases of a linear foreign body, which are often anchored at the pylorus in the dog, a gastrotomy is performed to release the proximal attachment, followed by enterotomy at the distal extent of the foreign body to allow complete extraction.
Closure is typically performed using 3-0 or 4-0 polydioxanone (PDS) in a single-layer, full-thickness pattern, either simple continuous (preferred by the author) or simple interrupted. For a right-handed surgeon, closure should proceed from right to left. When using a simple continuous pattern, the knots at the start and end of the suture line should be placed over intact serosa (i.e., not directly over the enterotomy site). Continuous tension should be maintained on the suture line throughout closure. This can be achieved by the surgeon or an assistant applying gentle traction to the suture during placement of each bite (Figure 6). Sutures should be placed approximately 3mm apart, incorporating 3mm of full-thickness intestinal wall, with particular emphasis on engagement of the submucosa. Additional simple interrupted sutures may be placed between continuous bites if required.
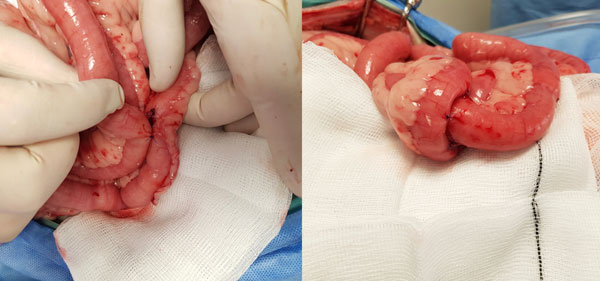
Figure 3: Intraoperative images demonstrating serosal patching of an enterotomy (left) and anastomotic site (right). The antimesenteric surface of the donor intestine has been tacked to each closure site with simple interrupted polydioxanone (PDS) sutures.
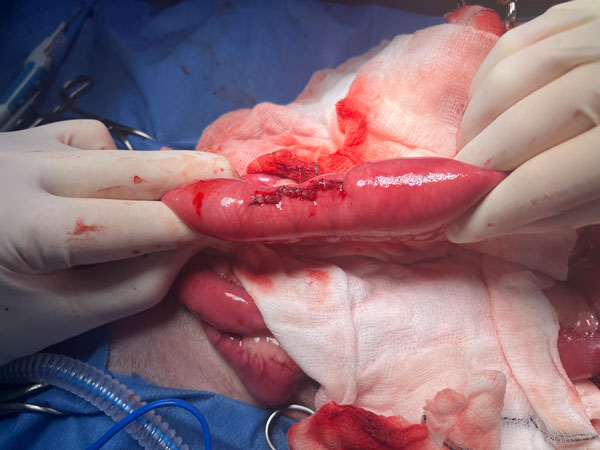
Figure 4: Intestinal leak testing in a dog. With both ends of the intestinal segment occluded using an assistant’s index and middle fingers, a 25-gauge hypodermic needle is inserted into the isolated lumen (not shown), and a volume of saline sufficient to achieve moderate distension is injected. The surgeon then gently compresses the distended segment while observing the suture line for leakage.
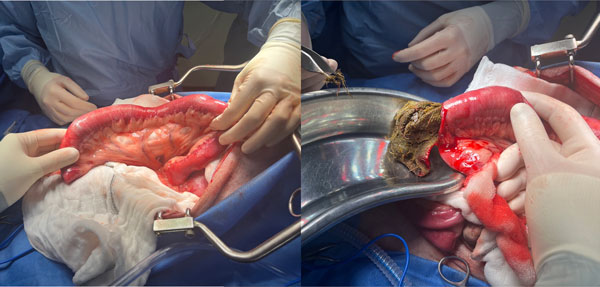
Figure 5: Intraoperative image of a sock foreign body that resulted in jejunal obstruction in a Labrador retriever. An enterotomy was performed aborally (distally) at the healthiest area of the affected jejunum. The sock was slowly and carefully removed into a kidney dish without contaminating the rest of the peritoneal cavity. Cranial to the right of the images.
Intestinal biopsy
For intestinal biopsy collection, a short longitudinal incision is made on the antimesenteric border, and a small full-thickness section of intestinal wall is excised using small, curved Metzenbaum or iris scissors, or a no. 11 blade. The incision is closed in a manner consistent with standard enterotomy technique, using a single-layer, full-thickness simple continuous or interrupted pattern with 3-0 or 4-0 polydioxanone (PDS).
An alternative technique involves placement of a stay suture on the antimesenteric surface of the intestine, and with gentle upward traction of the suture applied, a full-thickness section of intestine is excised along with the stay suture using scissors or a blade. A similar technique involves using the bent tip of a 25-gauge hypodermic needle to tent the antimesenteric surface of the intestine. Gentle, upward traction is applied via forceps attached to the needle, and a small full-thickness section of intestine is excised along with the needle (Figure 7).
Full-thickness intestinal biopsy using a 4-, 5-, or 6-mm skin biopsy punch has also been described. With this technique, stay sutures are placed on the antimesenteric surface on either side of the proposed biopsy site, and the biopsy punch is advanced into the intestinal lumen.
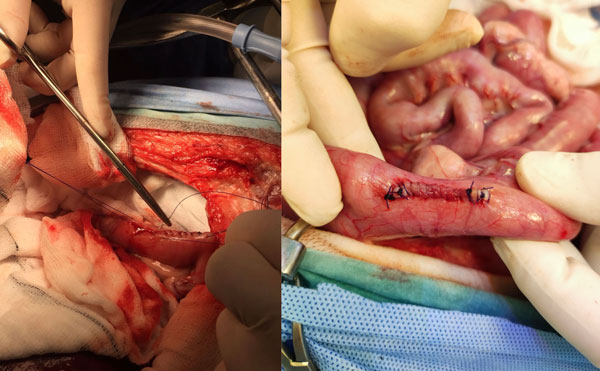
Figure 6: Intraoperative images demonstrating enterotomy closure in a dog using 3-0 polydioxanone (PDS) in a single-layer, full-thickness, simple continuous pattern. Stay sutures of 3-0 PDS have been placed to stabilise the intestinal segment (left). In the left image, an assistant maintains continuous tension on the suture line during placement of each bite. In the right image, additional simple interrupted sutures have been placed between continuous bites.
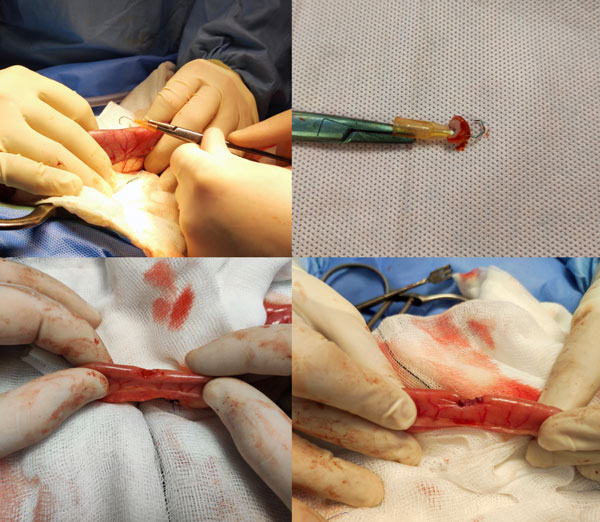
Figure 7: Intraoperative images demonstrating collection of a jejunal biopsy using the bent tip of a 25-gauge hypodermic needle to tent the antimesenteric surface of the intestine. Gentle traction is applied via forceps attached to the needle, and a small full-thickness section of intestine is excised en bloc with the needle. The biopsy site is closed in a manner consistent with a standard enterotomy, using 3-0 or 4-0 polydioxanone (PDS) in a single-layer, full-thickness, simple continuous or interrupted pattern.
Intestinal resection and anastomosis
Indications for intestinal resection and anastomosis include excision of ischaemic, necrotic, or neoplastic lesions, as well as the management of irreducible intussusceptions (Figure 8). After isolating the affected segment with moistened laparotomy sponges, the mesenteric vessels supplying the affected area are ligated with 3-0 polydioxanone and divided between ligature. The mesentery is incised near the vessels to be excised, leaving sufficient mesentery behind for later closure. Non-crushing intestinal forceps (e.g., Doyens) are placed at either end of the proposed enterectomy site. Alternatively, digital occlusion by an assistant’s index and middle fingers are used to prevent luminal contamination. Crushing forceps can be placed on the side of intestine being excised. The intestine is transected alongside the crushing forceps, leaving approximately 8-10mm of healthy intestine protruding beyond the Doyen forceps to allow easy suture placement for the anastomosis (Figure 9). A small arterial bleeder will often be present on the mesenteric side of the transected intestine and can be dealt with using bipolar electrosurgery or a tissue ligature. Anastomosis is performed using two simple continuous suture lines of 3-0 or 4-0 polydioxanone (PDS), one from mesenteric to antimesenteric and the other from antimesenteric to mesenteric, which are tied together. Suturing begins at the mesenteric border, where visualisation is often most challenging due to mesenteric fat. Cleaning the mesenteric border with a sterile, cotton-tipped bud/applicator can improve visualisation of the serosal surface and facilitate accurate suture placement (Figure 9).
Everted mucosa can be reduced by gently wiping it toward the lumen/cut surface with a moist cotton-tipped bud, or trimmed using Metzenbaum scissors. Sutures should be placed approximately 3mm apart, incorporating 3mm of the full-thickness intestinal wall, with particular emphasis on engagement of the submucosa (Figure 9). Simple interrupted closure may also be performed. Continuous tension should be maintained on the suture line throughout simple continuous closure. This can be achieved by the surgeon or an assistant applying gentle traction on the suture line during placement of each bite. Finally, the mesenteric defect is closed using 3-0 polydioxanone to prevent internal herniation, taking care to avoid mesenteric vessels supplying the small intestine.
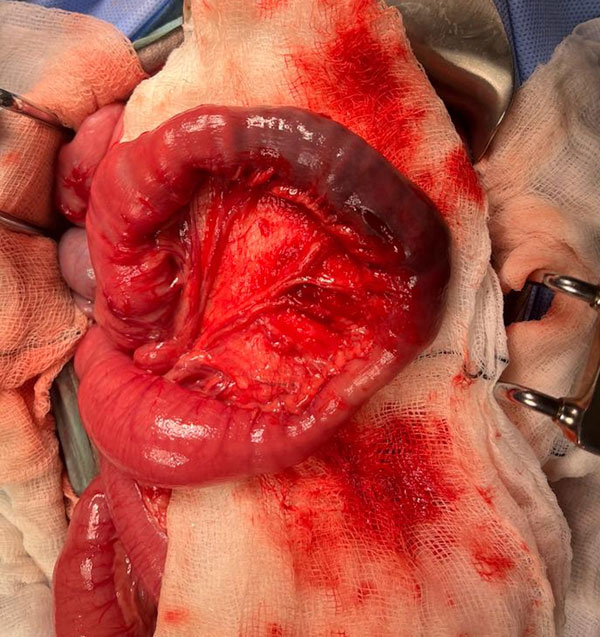
Figure 8: Intraoperative image of the jejunum in a dog demonstrating compromise of the intestinal wall following reduction of an intussusception.
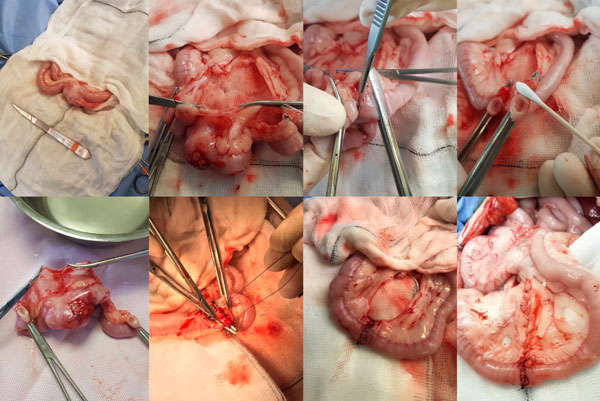
Figure 9: Series of intraoperative images demonstrating the steps involved in a small intestinal resection and anastomosis.
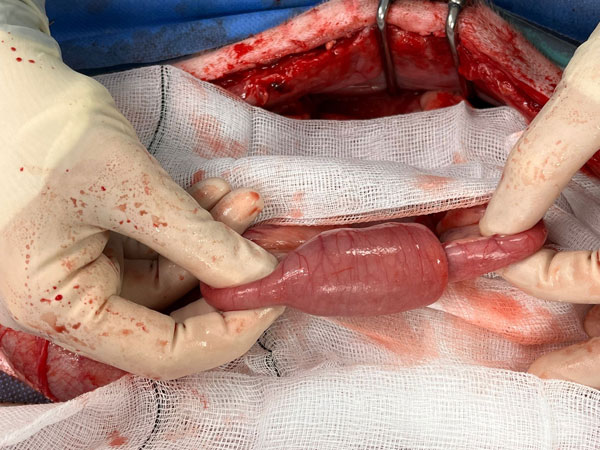
Figure 10: Intraoperative image demonstrating an intussusception in a dog, characterised by telescoping of a segment of small intestine into an adjacent segment, resulting in luminal obstruction and potential compromise of the mesenteric blood supply.
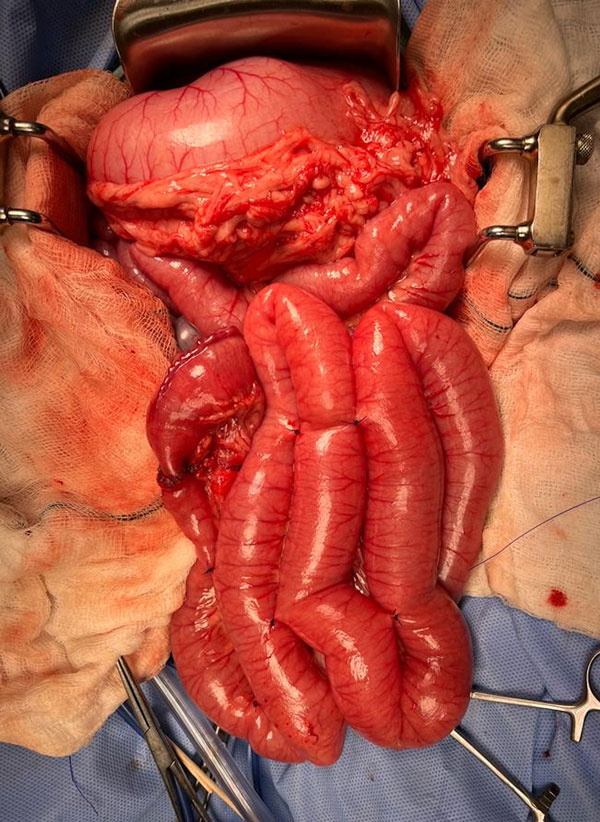
Figure 11: Intraoperative image demonstrating enteroplication of the small intestine. The affected segment is arranged into a series of gentle, parallel loops, avoiding kinks or sharp angulations that could compromise luminal flow. Adjacent intestinal loops are secured using absorbable, simple interrupted sutures.
Enteroplication
Intussusception involves invagination of one portion of the intestine (the intussusceptum) into the lumen of an adjoining segment (the intussuscipiens) (Figure 10). Enteroplication is a surgical technique used to reduce the risk of recurrent intussusception following manual reduction or resection and anastomosis. The procedure involves arranging the affected segment of small intestine into a series of gentle, parallel loops, ensuring there are no kinks or sharp angulations that could compromise luminal flow. Adjacent intestinal loops are then sutured together using absorbable, simple interrupted sutures of 3-0/4-0 polydioxanone (PDS) (Figure 11). These sutures are placed midway between the mesenteric and antimesenteric borders and incorporate the submucosa to provide adequate strength while minimising the risk of luminal narrowing. The aim is to prevent telescoping of the bowel, while preserving normal intestinal function. Although this technique may reduce recurrence, it is associated with potential complications including obstruction, perforation, and septic peritonitis. Recurrence is also possible at other sites, proximal or distal to the enteroplication. Enteroplication should be employed judiciously, as some studies have reported complication rates that exceed the risk of recurrence in cases managed without the procedure. As such, its use is not routine and should be considered on a case-by-case basis. It may be indicated in situations where the risk of recurrence is increased, including: when no underlying predisposing cause can be identified; when a causative factor cannot be immediately resolved (e.g., a resectable intestinal mass versus ongoing parasitism); in cases of recurrent intussusception; or where the owner may be unable to pursue repeat surgical intervention in the event of recurrence.
Conclusion
Small intestinal surgery remains a fundamental component of small animal surgical practice. Successful outcomes depend on careful patient stabilisation, meticulous surgical technique, and adherence to core principles of tissue handling and intestinal closure. Given the risk of serious complications, such as dehiscence and septic peritonitis, sound intraoperative decision-making and appropriate case selection are essential to optimise patient outcomes.
- Giuffrida MA, Cimino Brown D. Small Intestine. In: Johnston S, Tobias K, eds. Veterinary Surgery: Small Animal. 2nd ed. Elsevier Saunders; 2018:4671-4760.
- Allerton F, Sørensen TM, Scahill K, et al. European Network for Optimization of Veterinary Antimicrobial Therapy (ENOVAT) 2025 guidelines for surgical antimicrobial prophylaxis in dogs and cats. J Small Anim Pract. 2025 Dec 23.
- Grimes J, Schmiedt C, Milovancev M, Radlinsky M, Cornell K. Efficacy of serosal patching in dogs with septic peritonitis. J Am Anim Hosp Assoc. 2013;49(4):246-249.
- Mullen KM, Regier PJ, Fox-Alvarez WA, Case JB, Ellison GW, Colee J. Evaluation of intraoperative leak testing of small intestinal anastomoses performed by hand-sewn and stapled techniques in dogs: 131 cases (2008-2019). J Am Vet Med Assoc. 2021;258(9):991-998.
- Mullen KM, Regier PJ, Waln M, Colee J. Ex vivo comparison of leak testing of canine jejunal enterotomies: Saline infusion versus air insufflation. Vet Surg. 2021;50(6):1257-1266.
- Keats MM, Weeren R, Greenlee P, Evans KL, Minihan AC. Investigation of Keyes skin biopsy instrument for intestinal biopsy versus a standard biopsy technique. J Am Anim Hosp Assoc. 2004;40(5):405-10.
- Applewhite AA, Hawthorne JC, Cornell KK. Complications of enteroplication for the prevention of intussusception recurrence in dogs: 35 cases (1989-1999). J Am Vet Med Assoc. 2001;219(10):1415-8.
- Larose PC, Singh A, Giuffrida MA, Hayes G, Moyer JF, Grimes JA, Runge J, Curcillo C, Thomson CB, Mayhew PD, Bernstein R, Dominic C, Mankin KT, Regier P, Case JB, Arai S, Gatineau M, Liptak JM, Bruce C. Clinical findings and outcomes of 153 dogs surgically treated for intestinal intussusceptions. Vet Surg. 2020;49(5):870-878.
1. Which layer of the small intestine provides the primary strength for intestinal closure?
A. Mucosa
B. Submucosa
C. Muscularis
D. Serosa
2. Which of the following statements regarding antimicrobial use in small intestinal surgery is most appropriate?
A. Postoperative antibiotics are indicated for all enterotomies
B. Antibiotics are not required for clean-contaminated procedures
C. Postoperative antibiotics are not indicated in clean-contaminated procedures
D. Antibiotics should only be given after intestinal closure
3. When performing an enterotomy to remove an intestinal foreign body, the incision should ideally be made at which location?
A. Directly over the foreign body
B. Proximal to the obstruction
C. Distal (aboral) to the obstruction
D. At the mesenteric border
4. The small intestine receives its arterial blood supply primarily from which vessel?
A. Celiac artery
B. Caudal mesenteric artery
C. Cranial mesenteric artery
D. Portal vein
5. Surgery involving the duodenum is often more technically challenging than the jejunum. What is the primary reason for this?
A. The duodenum has a poorer blood supply
B. The duodenum is relatively fixed in position
C. The duodenum lacks a mesentery
D. The duodenum has a thicker muscular layer
ANSWERS: 1B; 2C; 3C; 4C; 5B.
















